Cattle infected with bTB eventually become weak, lose weight, get pneumonia and a chronic moist cough. Cows may decrease milk production. BTB is spread from infected animals via aerosol discharges and urine, saliva and manure contact. Cattle can become infected through sharing feeding and watering sites with infected cattle. Calves can become infected as well by consuming unpasteurized milk or colostrum with bTB. BTB can be transmitted between cattle to other warm-blooded animals and humans.
Fortunately, bTB-infected cattle are very rare in the U.S. This is the result of the National Tuberculosis Eradication Program, which started in 1917 when 5% of all cattle in the U.S. was infected with bTB. Nowadays, less than 0.006% of all cattle in the U.S. are infected with bTB. Virtually all bTB-infected cattle in the U.S. are located in a few counties in Michigan. In this region, the prevalence of bTB in local deer and other wildlife makes bTB difficult to eradicate.
The bTB Eradication Program, which includes the design of a surveillance and control protocol for bTB outbreaks, is administered by the USDA’s Animal and Plant Health Inspection Service (APHIS) in coordination with state animal health agencies. Because of this joint effort, bTB does not represent a serious threat to public or animal health in the U.S.
Running the bTB elimination protocol in an infected herd creates large costs to farms as well as to society by using taxpayers’ money.
The bTB elimination protocol can be roughly summarized as a series of whole-herd tests (WHT) performed to a herd infected with bTB. The initial screening for bTB-infected animals begins at slaughterhouse by meat inspectors. Any suspected animal at slaughterhouse is confirmed infected with lab tests (histology and culture tests). If an animal is confirmed infected by the lab test, the herd of origin is traced back and placed in quarantine, and the bTB elimination protocol begins.
First, a WHT is done to estimate bTB prevalence in the herd. This initial WHT consists of a caudal fold test (CFT) and, if positive, it is followed by a comparative cervical tuberculin (CCT) test. Both tests measure the immune response of the animal to bovine tuberculin proteins injected intradermally; in addition, CCT compares the immune response between bovine and avian tuberculin proteins. If the CCT test is positive, the animal is culled and tested with the lab tests described before. If the lab test is negative, the herd is considered free of bTB, and the quarantine is lifted, but if the lab test is positive, the protocol continues.
The rest of the protocol is divided into three phases: removal, verification and assurance testing. The removal test consists of a WHT of all animals 2 months old and older with a CFT test. This WHT is repeated at least 60 days apart until two consecutive negative WHTs are achieved (no animals test positive). At this point, the removal phase is over, and the verification phase begins. More than two consecutive negative WHTs may be required to proceed to the next phase if the prevalence is deemed high.
In the verification phase, all animals over 6 months old are tested. The verification test begins 180 days after the last removal test and consists of a CFT test and, if positive, a CCT test. If all CCT tests are negative, the quarantine is lifted. The assurance test is conducted after one year of the quarantine release. It requires the same tests as the verification test but applied to animals older than 12 months.
The bTB protocol starts from the beginning, at the removal phase, if any animal is confirmed bTB-positive by lab tests at any stage of the protocol.
Farmers are compensated for all culled animals at market value. Because the CFT and CCT tests are not perfect, many healthy animals are culled due to false positive results. Farmer suffer lost revenue of milk production; thus, indemnity payments may underestimate actual losses by the farm.
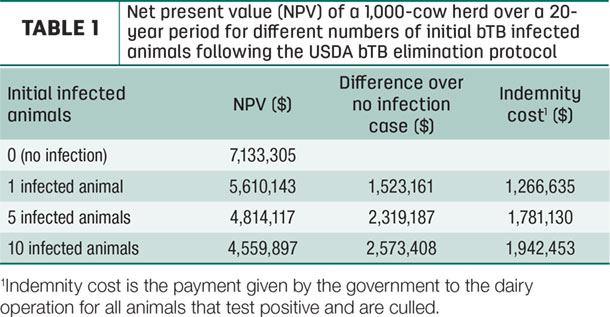
In a recent analysis, we estimated the total costs of performing the bTB elimination protocol in a 1,000-cow dairy herd when infected animals are introduced in the herd. We found that introducing one bTB-infected animal creates costs of about $1.5 million, out of which about $1.3 million (83 percent) are indemnity costs. The value of a healthy 1,000- cow herd is estimated at $7.1 million. Introducing five and 10 bTB-infected animals into the herd results in costs of $2.3 and $2.6 million, with corresponding indemnity costs of $1.8 and $1.9 million, respectively. The costs are equivalent to 21%, 32% and 36% of the herd value for one, five and 10 bTB-infected animals introduced in the herd, respectively. Results are shown in Table 1.
We estimated that detecting bTB in our simulated dairy herd can take more than 900 days after the introduction of one bTB-infected animal in the herd; by the time of detection, on average, four infected animals remain in the herd.
Even though the risk of bTB infection is very low – in 2017, only 13 animals were detected at slaughter in the U.S. – the cost of running the protocol can be substantial. Farmers can minimize their risk of infection by purchasing cattle originated from bTB-negative farms or regions. Cattle of foreign origin are at higher risk than domestic cattle, but almost all of them are destined to the beef market and end up in feedlots.
Despite the high costs, the protocol is effective and efficient in eliminating bTB-infected animals from a herd. The cost depends mostly on current milk margins and on the quality of the test results. Improved test quality (sensitivity and specificity) would decrease the costs of the protocol considerably. ![]()
References omitted but are available upon request. Click here to email an editor.
Leslie J. Verteramo Chiu is a research associate with the department of population medicine and diagnostic sciences at the Cornell University College of Veterinary Medicine. Email Leslie J. Verteramo Chiu.





