Dairy owners, veterinarians, herd managers and employees play a significant day-to-day role in food safety. Antibiotic residues in milk and dairy beef are an important food safety issue, as antibiotics may cause severe allergic reactions in some people. Milk All farm bulk tank milk and tanker truck milk must be tested for evidence of antibiotic residue prior to processing, according to the Pasteurized Milk Ordinance of the Food and Drug Administration (FDA). Milk found with antibiotic residues is dumped, with the dairy producer bearing the cost for the dumped milk.
The National Milk Drug Residue Database is operated by an independent third party under contract with the FDA. According to the database report for 2009, 0.026 percent of all samples tested were positive for a drug residue, down from 0.104 percent in 1996 (Table 1) .
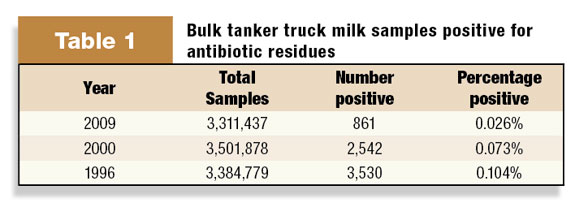
Although the database was designed to promote maximum participation by the dairy industry to voluntarily report all testing results, it is important to recognize that the samples reported do not necessarily represent 100 percent of the milk supply from each state.
Nevertheless, the downward trend over the past 14 years is great news and is most likely related to successful teamwork among dairy owners, veterinarians, herd managers, employees, allied industry technical support, milk processors and university faculty.
Dairy beef
Dairy cows represent a major source of beef. In recent years, meat packers have sharpened their focus on the quality of cattle coming into the packing plant. Inspectors from the USDA Food Safety and Inspection Service (FSIS) use both ante-mortem (before death) and post-mortem (after death) findings as bases for selecting cattle for residue testing.
Common ante-mortem conditions associated with a high violation risk include evidence of recent surgery, and slow, weak or dehydrated animals. Post-mortem conditions associated with increased violation risk include: mastitis, metritis, injection site lesions and abomasal disease. Only 15 percent of violations result from ante-mortem findings, providing evidence that comprehensive post-mortem inspections are successful in the identification of violative residues.
According to a large U.S. slaughter facility that processes market dairy cows, approximately 5 percent to 15 percent of the slaughter population is tested annually, depending on the quality of cattle. Furthermore, 100 percent of cattle exhibiting targeted ante-mortem and post-mortem conditions (according to the meat packer’s Hazard Analysis Critical Control Point plan), have been tested since 1995.
Carcasses found to be positive on the initial test for drug residues are retained pending further laboratory tests on kidney, liver and muscle tissues. Violators are notified within a few days of laboratory confirmation, and an explanation for the cause of the violation is obtained. Then, the producer must provide evidence of immediate corrective action and long-term preventive measures. The cost of the residue-condemned carcass is billed to the producer, with the warning that repeat violations will result in placement on the USDA-FSIS Residue Violator Alert List, and possible market loss.
The good news is that drug residues in dairy beef carcasses are decreasing. At the slaughter facility mentioned previously, there were 248 violative carcasses in 2007 (the most recent year for which data is available), down from 459 reported at the same meat-packing plant for the year 2000.
According to the FDA, failure to observe label withdrawal periods before slaughter is the principal cause of illegal drug residues. Another major cause of violative residues is failure to observe label directions regarding dosage and route of administration.
Educational resources
There are many tools available to help avoid antibiotic residues in milk and dairy beef. These tools include:
• “Milk and dairy beef residue prevention protocol – 10 point plan”: Available in English at www.agri-ed.com/catalog.html . Key segments of the manual include questions designed to help producers identify areas which may cause drug residue problems, a list of FDA-approved drugs for lactating and non-lactating cows, and a list of milk screening tests for detecting residues in bulk tank milk.
• “Idaho Dairy Beef Quality Assurance” book: The book was developed by a University of Idaho Extension team led by Jason Ahola (beef) and Mireille Chahine (dairy) to help dairy producers educate their employees about the importance of beef from market dairy cows. To request a copy of the English or Spanish version, send an e-mail to: beef@uidaho.edu
• “Dairybeef: Maximizing quality and profits” website: This website ( http://dairybeef.ucdavis.edu ) contains videos, narrated slide sets, and on-farm tools regarding meat safety and quality. In addition, key segments have been translated and are available for Spanish-speaking farm employees.
Food safety remains an important issue for producers and consumers, even with the downward trend in milk and dairy beef samples positive for antibiotic residues. Consequently, food safety continues to be a major focus of the USDA’s National Institute of Food and Agriculture through federal funding for research, education and extension programs. For more information regarding residue avoidance in milk and dairy beef, contact your veterinarian, local cooperative extension or milk processor to help implement a strategy to avoid violative residues in milk and dairy beef. PD
PHOTO: All farm bulk tank milk and tanker truck milk must be tested for evidence of antibiotic residue prior to processing, according to the Pasteurized Milk Ordinance of the Food and Drug Administration (FDA). Photo by PD staff.

-
Joseph C. Dalton
- Dairy Specialist
- University of Idaho
- Email Joseph C. Dalton






